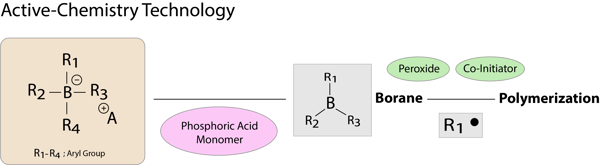
Active-Chemistry is superior to the ocnventional chemical polymerization initiator, a benzoyl peroxide/amine system, because it exhibits high catalytic activity under strongly acidic conditions. A thin bonding layer formed after air blow becomes hard because of rapid progression of polymerization and curing on its adhesive interface (self-cure), when it comes into contact with resin materials such as composite resin.
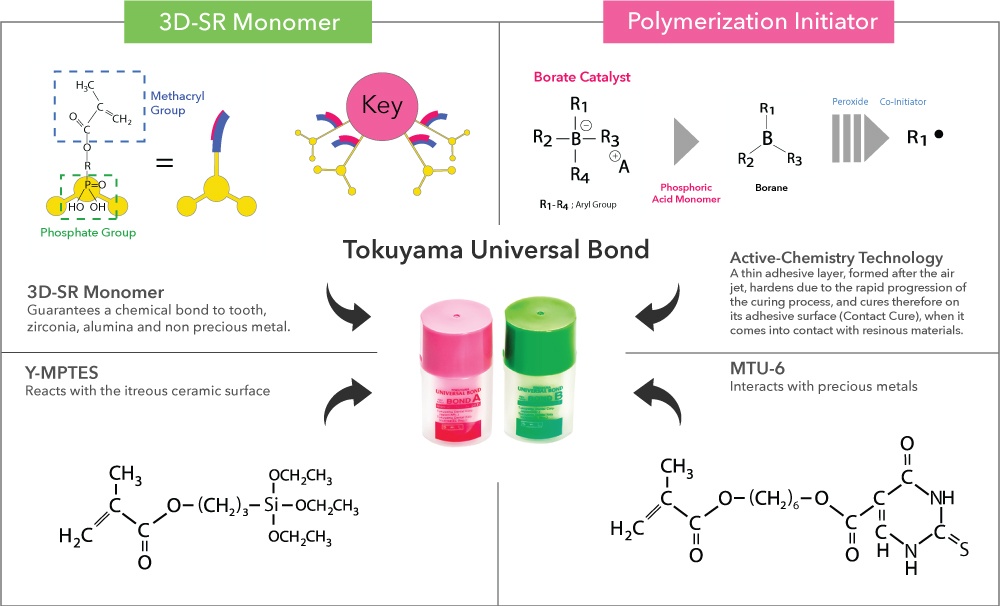
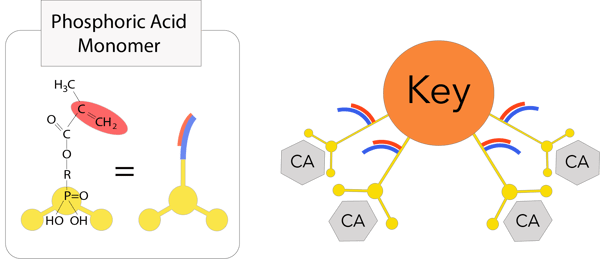
The 3D-SR monomer have several functional groups that can interact with calcium and polymerizing groups per molecule. Tokuyama UNIVERSAL BOND has an enhanced response to tooth calcium and durability by using a new 3rd generation 3D-SR monomer.

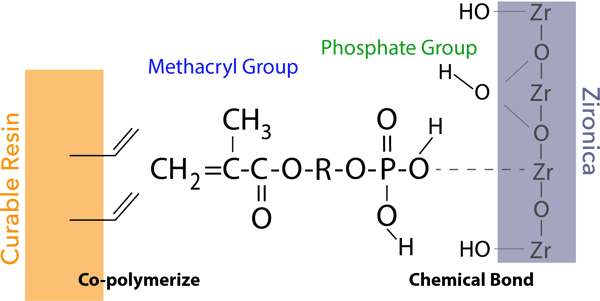
The phosphate group of the new 3D-SR monomer forms chemical bonds with the zirconia/alumina surface for adhesion.
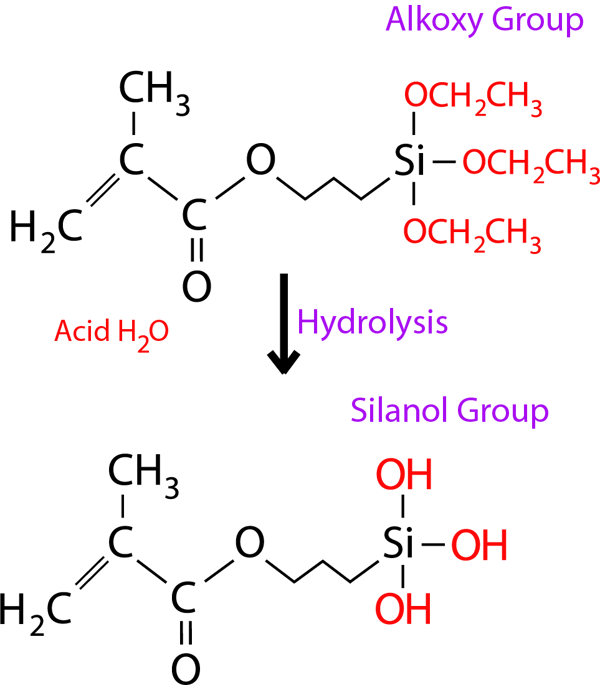
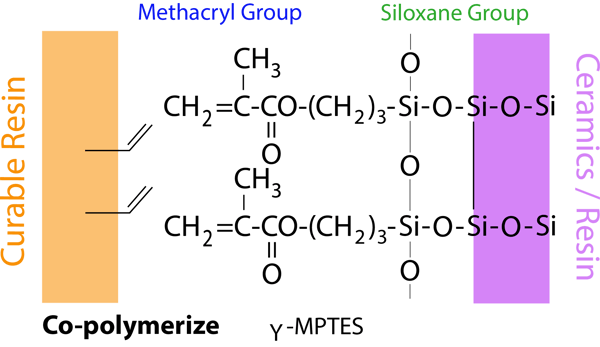
The alkoxy group in γ-MPTES reacts with water to form a silanol group and next, a siloxane bond is formed by a dehydration and condenstation reaction with the silanol group on the ceramic surface. Additionally, the methacryl group co-polymerizes with monomers in dental curable materials. Since the new silane coupling agent, γ-MPTES is more stable in the bottle than the conventional one (γ-MPS), the adhesion effect lasts for a long time.
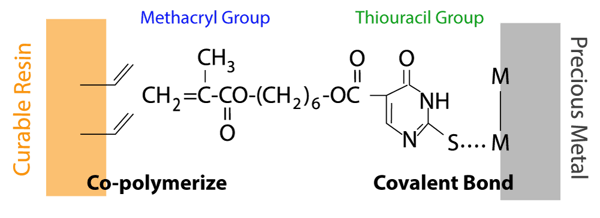
The sulfur atom in the thiouracil group of MTU-6 interacts with precious metal (covalent bond) and additionally, the methacryl group co-polymerizes with monomers in dental-curable materials.
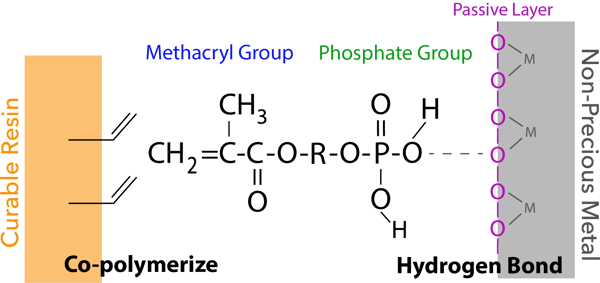
The phosphate group of new 3D-SR monomer interacts with the oxygen atom of the passive layer of a non-precious metal surface (hydrogen bond) and additionally, the methacryl group co-polymerizes with monomers in dental curable materials.