In regard to bonding agents, the term “universal” is not regulated and can mean different things from product to product. The Dental Advisor has defined three criteria, and for a bonding agent to be considered “universal”, it has to satisfy at least two of them. Dental Advisor. May-June 2017 Vol. 34, No.3
Tokuyama Universal Bond meets all three criteria, making it a “true universal” without the use of extra products:
- Self-etch; compatible with total-etch and selective-etch techniques
- Compatible with all light-cure, dual-cure, and self-cure materials without use of activators
- Can be used as primer for silica-based, zirconia-based, and metallic restorations
Indications

Universal Use Comparison Table
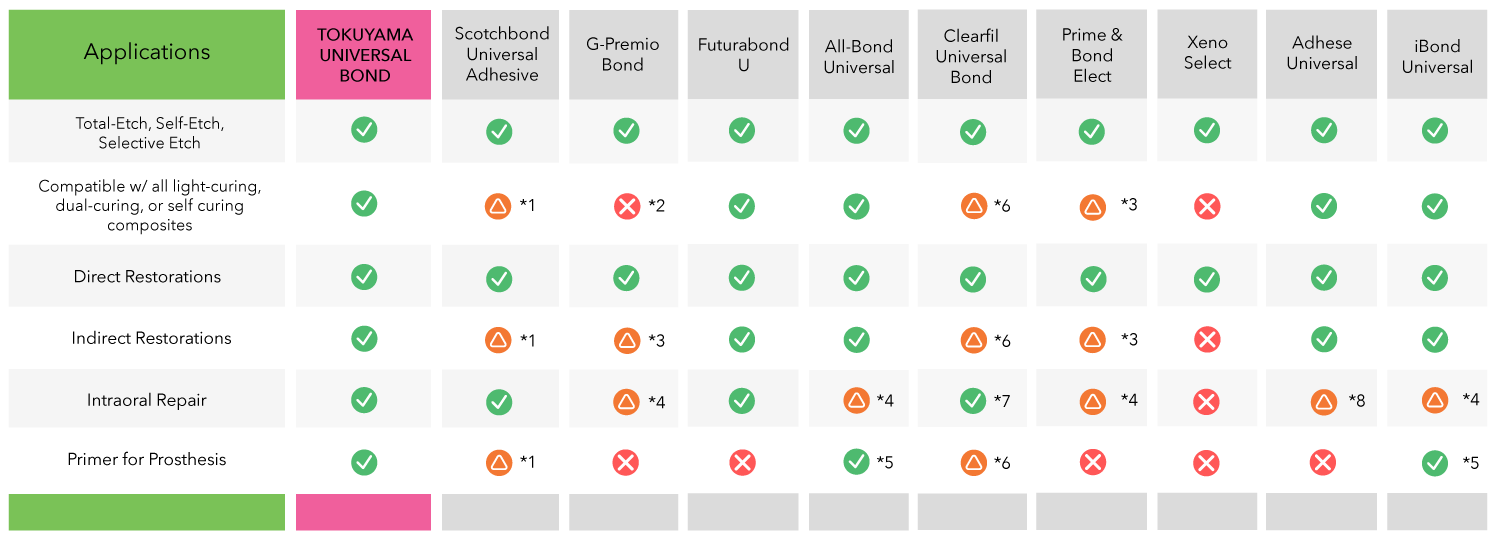
- *1 Requires Dual Cure Activator (DCA) unless it is used with Rely X Ultimate
- *2 Bonding of dual-cured core build up composites to tooth structure as long as these materials are light-cured
- *3 Requires DCA
- *4 Requires Primer
- *5 Requires light-curing
- 6 Requires DCA and light-curing unless it is used with CLEARFIL DC CORE PLUS or PANAVIA SA CEMENT
- *7 Primer recommended
- *8 Only composite repair
Tokuyama Universal Bond offers high bond strength to polymerizable resin materials (resin cement, acrylic resin, and composite resin) and indirect restorative materials such as glass-ceramics (porcelain), oxide-ceramics (zirconia and alumina), metals (precious and non-precious), and resin materials including inorganic filler.
Shear Bond Strength Comparison Charts
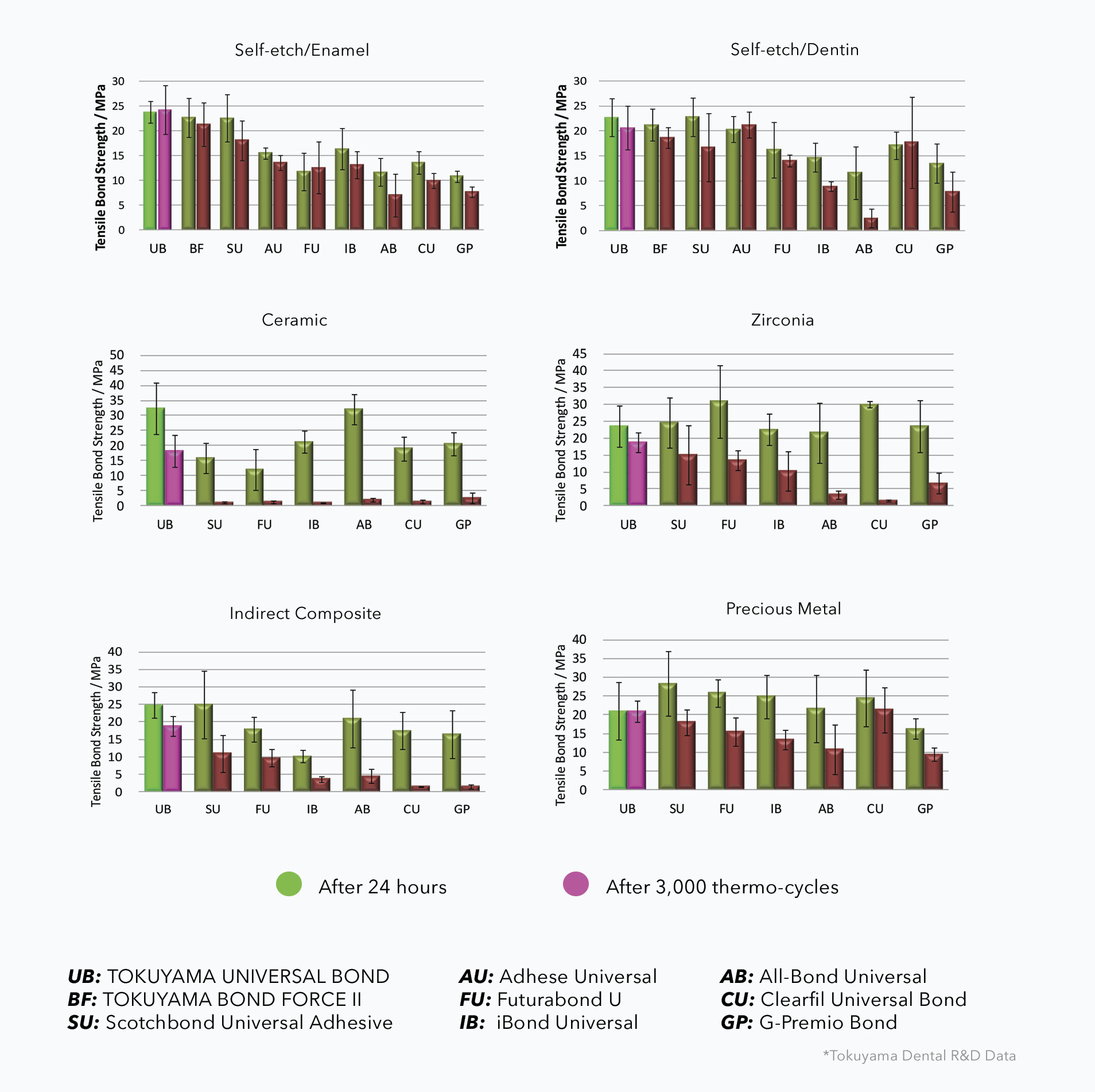
*The 35th Annual Meeting of Japan Society for Adhesive Dentistry, 2016
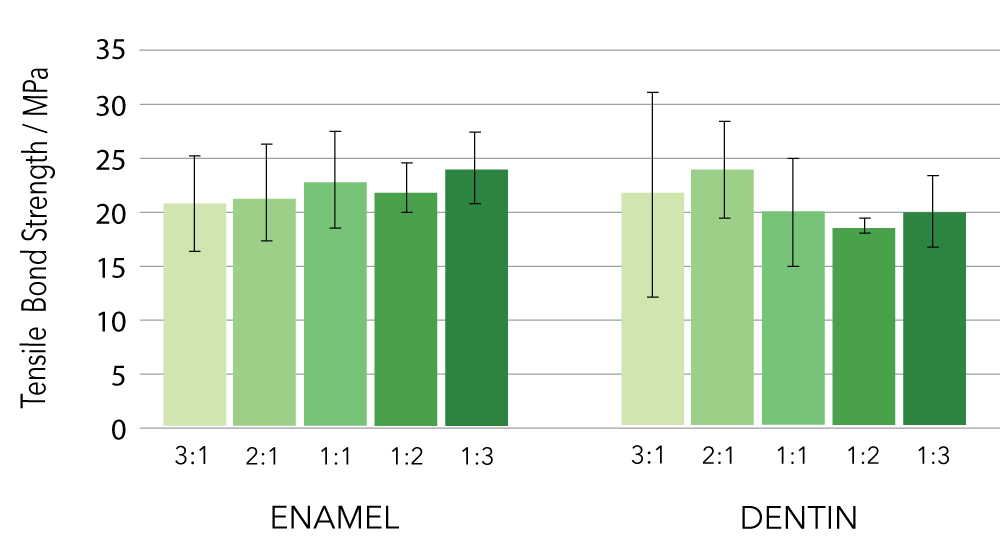
Mixing Ratios
The recommended mixing ratio for Tokuyama Universal Bond is 1:1 Bond A & B, but it maintains performance within a mixing ration of up to 1:3, leaving room for error:
Cavity Adaptation – Direct Restoration
Excellent cavity adaptation provides void-free bonding.

*Tokuyama Dental R&D Data
Reduce chair time, cost, and inventory by eliminating the need to add activators or primers, agitate surfaces, light-cure, or wait after placement. Follow the same three quick and easy steps for any material used in direct and indirect restorations:

Protocol Timeline Comparison for Direct Restorations
Within 25 seconds, Tokuyama Universal Bond delivers the quickest & easiest application protocol for every case and material
Self-cure chemistry provides reliable bond in deep preparations, as well as post and core build-ups that are difficult to reach with a curing light.
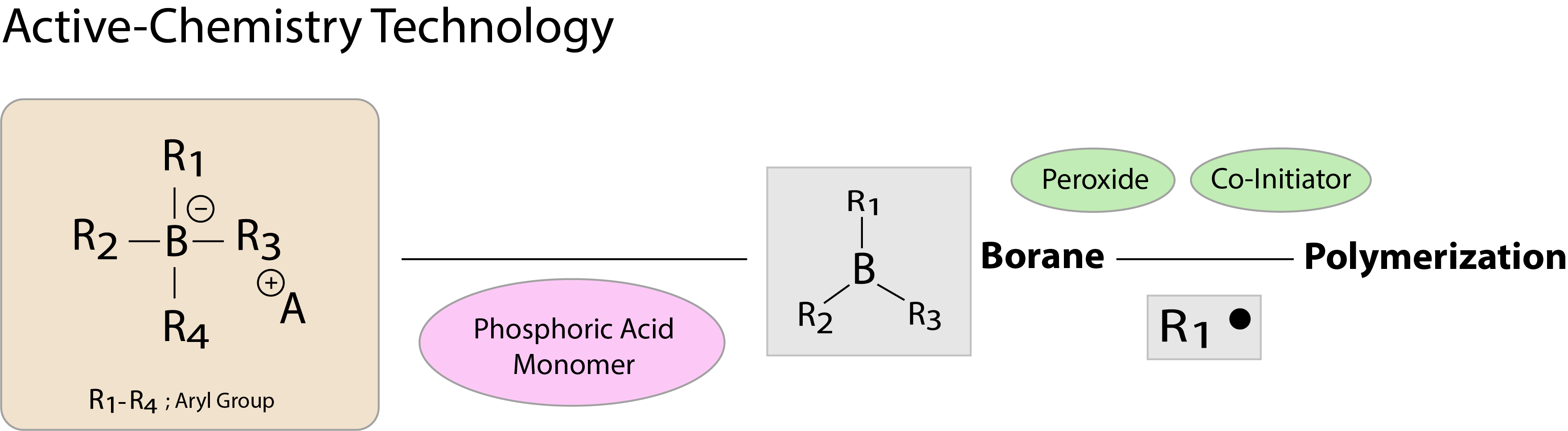
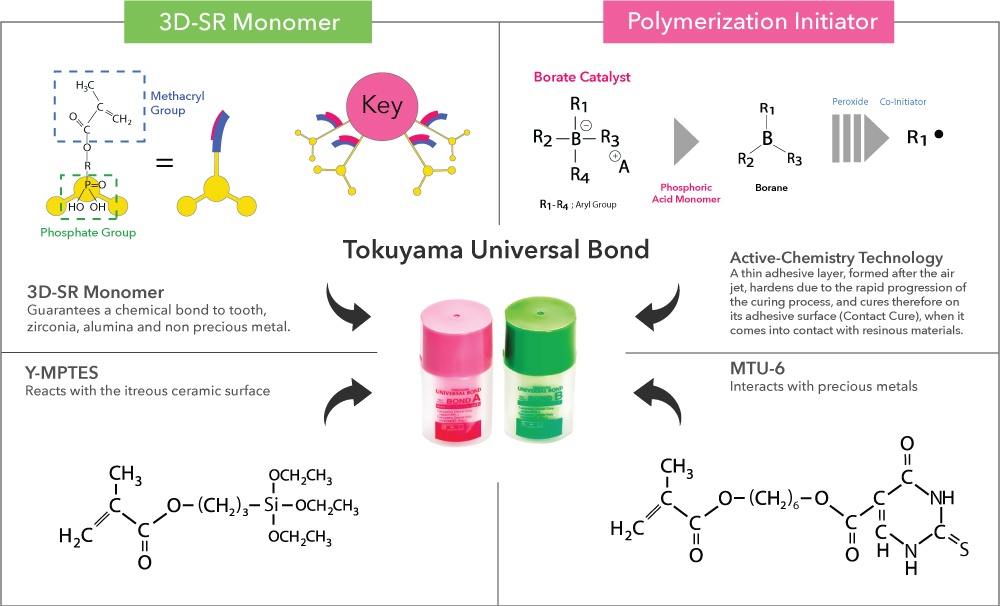
Active-Chemistry technology is superior to the conventional chemical polymerization initiator, a benzoyl peroxide/amine system because it exhibits high catalytic activity under strongly acidic conditions. A thin bonding layer forms after exposure to air, and hardens because of rapid progression of polymerization and curing on its adhesive interface (self-cure) once it comes into contact with resin materials such as composite.
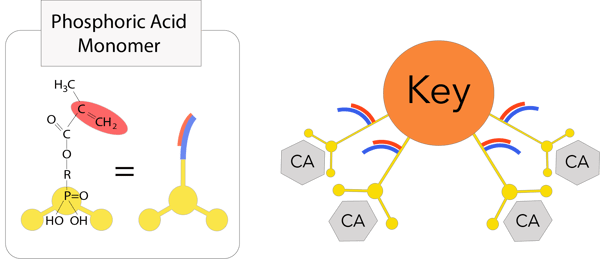
3rd Generation 3D-SR Monomer
3rd Generation 3D-SR Monomer The 3D-SR monomer has several functional groups that can interact with calcium and polymerizing groups per molecule. Tokuyama Universal Bond has an enhanced response to tooth calcium and durability by using a new 3rd generation 3D-SR monomer.
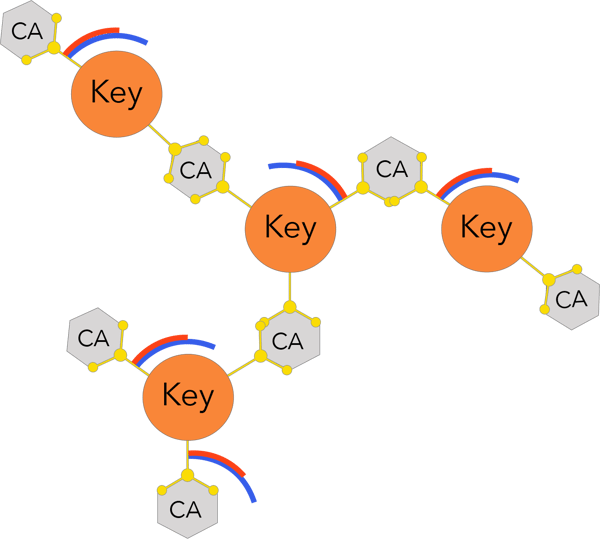
- Strong bonding to tooth structure through multi-point bounding.
- Improved adhesion layer strength through three-dimensional cross-linking reactions.
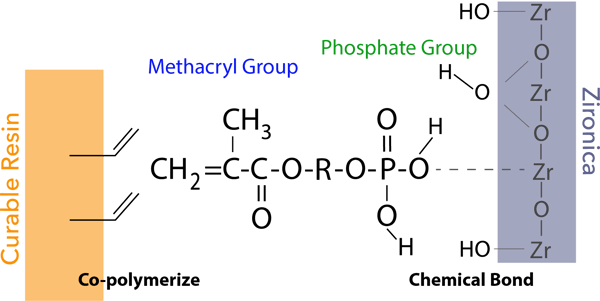
The phosphate group of the new 3D-SR monomer forms chemical bonds with the zirconia/alumina surface for adhesion.
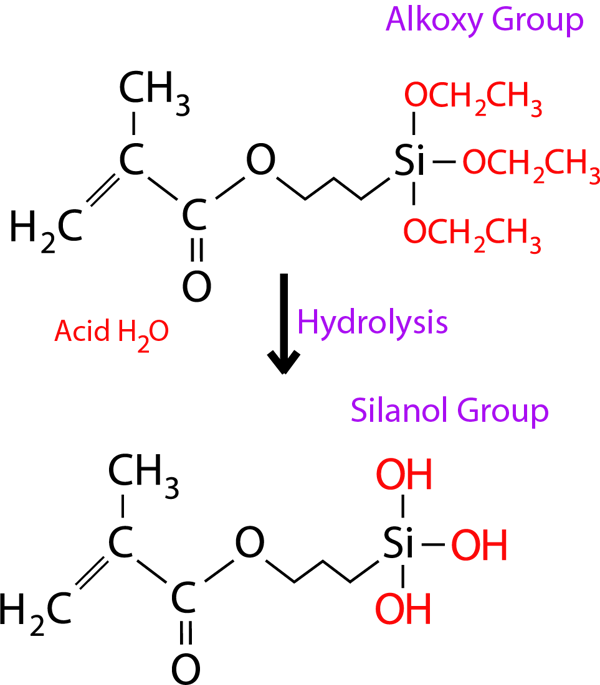
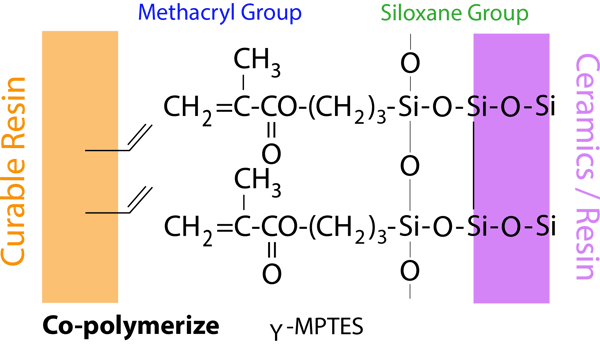
The alkoxy group in γ-MPTES reacts with water to form a silanol group. Next, a siloxane bond is formed by a dehydration and condensation reaction with the silanol group on the ceramic surface. Additionally, the methacryl group co-polymerizes with monomers in dental curable materials. Since the new silane coupling agent, γ-MPTES, is more stable in the bottle than the conventional one (γ-MPS), the adhesion effect lasts for a long time.
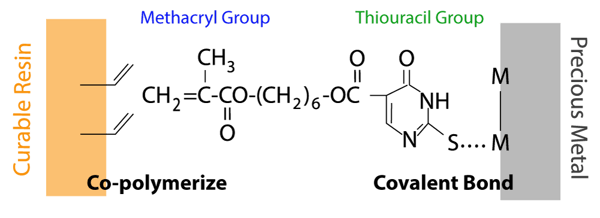
The sulfur atom in the thiouracil group of MTU-6 interacts with precious metal (covalent bond) and additionally, the methacryl group co-polymerizes with monomers in dental-curable materials (resin cements, bonding agents, resin composites, etc.).
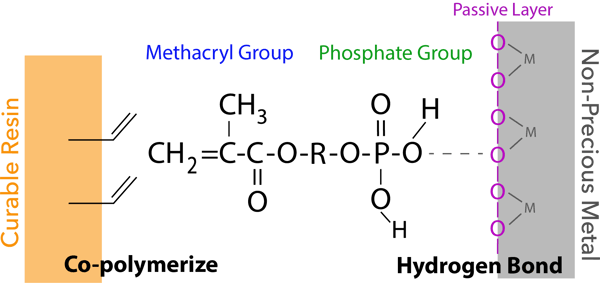
The phosphate group of new 3D-SR monomer interacts to the oxygen atom from the passive layer of a non-precious metal surface (hydrogen bond). Additionally, the methacryl group co-polymerizes with monomers in dental curable materials (resin cements, bonding agents, resin composites, etc.).
Put some clear and concise content in here. Keep your reader in mind. What are they trying to do? What are they hoping to learn? Why are they reading this? Help them out and use the tone and voice of your organization.
Put some clear and concise content in here. Keep your reader in mind. What are they trying to do? What are they hoping to learn? Why are they reading this? Help them out and use the tone and voice of your organization.
Put some clear and concise content in here. Keep your reader in mind. What are they trying to do? What are they hoping to learn? Why are they reading this? Help them out and use the tone and voice of your organization.